Teaming for Interdisciplinary Research Pre-Seed Program
Tumor-Specific Neoantigen Prediction
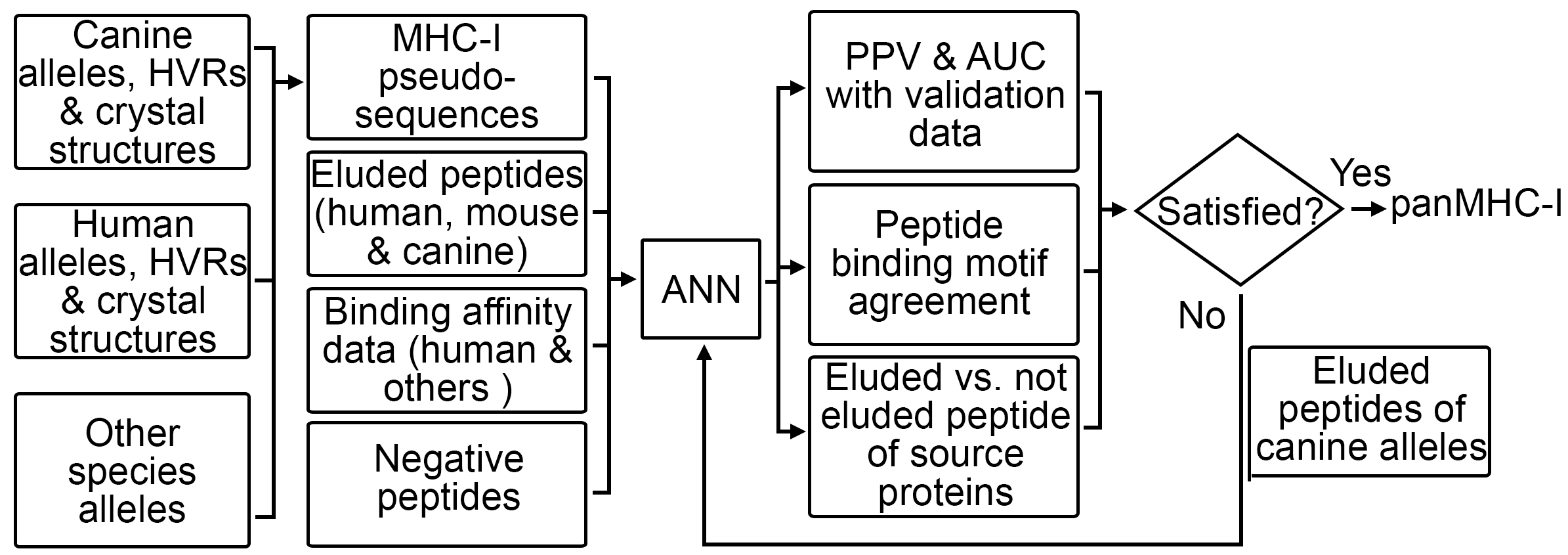
Immunotherapy holds great promise in cancer treatment, as it harnesses the patient’s immune system to specifically target and kill tumor cells. However, significant challenges prevent the full realization of its great potential. One of these challenges is that many promising preclinical results have failed to translate into clinical success, due to a gap between preclinical models and human clinical trials. Spontaneous cancers in pet dogs have the potential to effectively bridge this gap, being naturally occurring and with an intact immune system.
However, the lack of essential resources creates roadblocks to the effective use of canine cancers. The deficiency is clearly seen in systematic tumor-specific neoantigen (TSNA) prediction. First, cancer somatic mutation discovery represents the 1st step of TSNA prediction. Its accuracy however is compromised by the current canine gene annotation, which is far behind its human counterpart in terms of accuracy and comprehensiveness. Second, there are no software tools available for canine major histocompatibility complex class I (MHC-I) genotyping with next-generation sequencing (NGS) data, the 2nd step of TSNA prediction. The human genotyping tools do not work for the dog, due to the deficiency in known canine MHC-I alleles. Lastly, there lack tools for canine TSNA prediction, and current tools developed for human cancers have a low prediction accuracy for the dog.
With NGS data published for thousands of dogs from hundreds of breeds, now is the time to address these deficiencies. We propose to harness these massive NGS data and our >14 years’ experience in comparative canine and human cancer research to: 1) improve canine gene annotation by harnessing public canine RNAseq and other data, and by building comprehensive dog-human molecular links; 2) develop software tools for MHC-I genotyping and TSNA prediction for the dog; and 3) disseminate our tools and data resource effectively to the canine community. Our proposed study will significantly speed up the development of cancer immunotherapy. Our work will contribute to the development of the Integrated Canine Data Commons, a database newly initiated by the National Cancer Institute to disseminate canine molecular and clinical data to the public. Lastly, our research will contribute to informatics technology, by developing innovative strategies for inter-species big data integration.
Team Lead
Shaying Zhao
Department of Biochemistry and Molecular Biology
szhao@uga.edu
Team Members
Stephen M. Tompkins
Center for Vaccines and Immunology
Kevin Dobbin
Department of Epidemiology and Biostatistics
Tianming Liu
CSCI Computer Science
Robert Woods
Complex Carbohydrate Research Center
Corey Saba
Department of Small Animal Medicine
Kristina Meichner
Department of Pathology
